Save costs with our PCR Master Mixes
Master mixes help you to save time and reproducibility
Our ready to use Master Mixes are great alternative options to your regular individual PCR reagents. Master mixes are reagents that include all the components needed for a PCR. By only the extra step of adding your template and primer pairs you are able to run a PCR quickly and successfully.
- Minimize possible contamination of the stock reagents due to fewer handling steps, enhancing the reproducibility of your experiments.
- Reduce your experiments time.
Share your research, challenges and experiences with us so we can so we can discuss the best approach on how to proceed with your research.
We offer a broad range of master mixes. Have a look at the ones described below to choose which ones match better with your project needs.
- Taq DNA Polymerase Master Mix RED
- RealQ Plus PCR Master Mix
- NEW! RealQ Fast 2x Master Mix Green
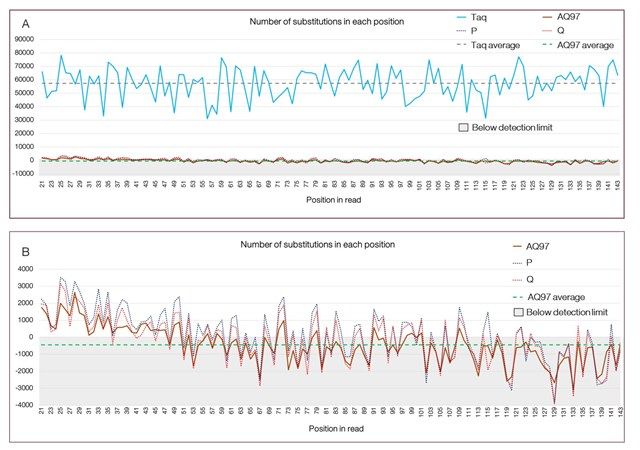
- AQ97 High Fidelity DNA Polymerase Master Mix
- Other Master Mixes
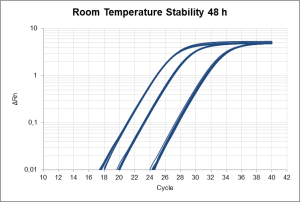
Each of our Master Mixes has its own benefits. All the master mixes specified below are available with different buffer options, giving wider options to finally chose the master mix that matches your PCR conditions and application.